The Strategy for the prioritisation of substances under HBM4EU was developed in the first half of 2017 and agreed at the meeting of the Governing Board in September 2017.
The strategy sets out the steps involved in identifying substances for research and surveys under the project. The Strategy also includes a set of prioritisation criteria, against which substances were assessed.
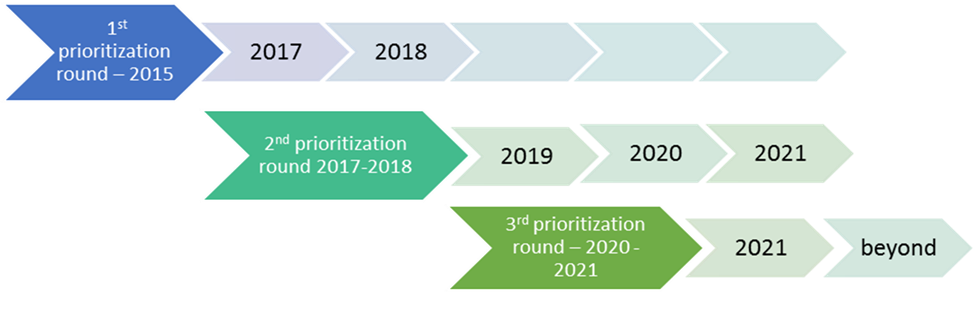
Throughout the duration of HBM4EU (2017-2021), three prioritisation rounds took place as shown below.
Prioritisation rounds in HBM4EU
- The 1st prioritisation process took place in 2015
- The 2nd one between 2017-2018
- The 3rd one between 2020-2021
Disclaimer
The HBM4EU project was launched in 2016 with the aim of improving the collective understanding of human exposure to hazardous chemicals and developing HBM as an exposure assessment method. The project had €74m in funding and jointly implemented by 120 partners from 28 participating countries – 24 EU member states plus Norway, Switzerland, Iceland and Israel and the European Environment Agency. One of its aims was to ensure the sustainability of HBM in the EU beyond 2021. The project ended in June 2022. The website will not be updated any longer, except the page on peer reviewed publications, but will be online until 2032.