The Chemicals Strategy for Sustainability – how human biomonitoring contributes
by Cristina De Avila, Head of Unit on Sustainable Chemicals, Directorate-General for Environment, European Commission
The European Commission adopted the EU Chemicals Strategy for Sustainability in October last year, as the first delivery of the Zero Pollution Ambition announced in the Green Deal. The objective of the Strategy is to boost innovation for safe and sustainable chemicals, and to increase the protection of human health and the environment against hazardous chemicals.
The Chemicals Strategy recognises the fundamental role of chemicals for human well-being and for the green and digital transition of the European economy and society. At the same time, it acknowledges the urgent need to address the health and environmental challenges caused by the most harmful chemicals. It lists concrete actions in five chapters.
To increase the protection of health and the environment, flagship initiatives include in particular:
- Phasing out the most harmful substances from consumer products, such as toys, childcare articles, cosmetics, detergents, food contact materials and textiles, unless their use is proven essential for society. Those most harmful substances include among others endocrine disruptors, chemicals that affect the immune and respiratory systems, and persistent substances such as per- and polyfluoroalkyl substances (PFAS);
- Minimising and substituting as far possible the presence of substances of concern in all products. Priority will be given to those product categories that affect vulnerable populations and those with the highest potential for circular economy;
- Addressing the combination effect of chemicals (cocktail effect) by taking better account of the risk that is posed to human health and the environment by daily exposure to a wide mix of chemicals from different sources;
- Ensuring that producers and consumers have access to information on chemical content and safe use, by introducing information requirements in the context of the Sustainable Product Policy Initiative.
To boost innovation and competitiveness of the EU industry producing and using chemicals, the strategy aims to make chemicals safer and more sustainable by design. This is not only an urgent necessity but also a great economic opportunity. The Strategy will support industrial innovation through a number of actions, so that such chemicals become the norm on the EU market and a benchmark worldwide. This will be done mainly by:
- Developing safe-and-sustainable-by-design criteria and a dedicated EU-wide support network;
- Ensuring the development and uptake of safe and sustainable-by-design substances, materials and products through EU funding and investment instruments and public-private partnerships;
- Direct funding to fill knowledge gaps on the impact of chemicals, promote innovation and move away from animal testing;
- Stepping up enforcement of EU rules both at the borders and in the single market;
- Simplifying and consolidating the EU legal framework – e.g. by introducing the ‘One substance one assessment’ process, strengthening the principles of ‘no data, no market’ and introducing targeted amendments to REACH and sectorial legislation, to name a few.
The Strategy emphasises the importance of a comprehensive knowledge base on chemicals, and stresses the importance of human, but also environmental (bio)monitoring. To guarantee such activities, it announces the continuation of financial support for EU-wide activities in this field. Further actions in this area include
- The establishment of a research and innovation agenda for chemicals, driven by a EU-level Coordination Group, that would also promote the regulatory uptake of research findings;
- Fostering multidisciplinary research and digital innovations for advanced tools, methods and models, and data analysis capacities to also move away from animal testing;
- The development of an EU early warning and action system for chemicals to ensure that EU policies address emerging chemical risks as soon as identified by monitoring and research,
- The development of a framework of indicators to monitor the drivers and impacts of chemical pollution and to measure the effectiveness of chemicals legislation.
The Strategy also contains a list of actions to consolidate and simplify the EU legal framework, and to ensure that the EU sets the example for a global sound management of chemicals.
As soon as the Strategy was adopted, we started implementing it. Our plans are diverse. Not only do we offer financial support to researchers, innovators and industry to enable the green and digital transition, but also we are revising the two cornerstones of the EU regulatory framework on chemicals, the REACH and CLP regulations. Those revisions will happen in a targeted way, focused on achieving the objectives outlined in the Strategy. They will be based on impact assessments and will include public consultations. The vision and objectives of the Strategy are very ambitious; we will only succeed in achieving them through a joint effort of all relevant societal and economic actors to move together towards a toxic-free environment.
Sustainability of HBM4EU: the iceberg, the cocktail and the orchestra
by Professor Robert Barouki, French National Institute of Health and Medical Research (INSERM), Leader of HBM4EU Pillar 3 on exposure and health
European citizens are extremely concerned by the presence of thousands of chemicals in their environment and in consumer products. They feel that they have the right to know what they are exposed to, which chemicals they have in their body and how they should interpret this using the best available science. Informing policy makers and providing science-based information for the public debate is key to our initiative.
Last September, the Management Board of HBM4EU sent a letter to the Vice President of the European Commission and to the Commissioners of relevant Directorate-Generals urging them to support sustained human biomonitoring activities in the EU, in line with the objectives of the European Green Deal. The following text is inspired by the content of this letter with some highlights on the major objectives of the Chemicals Strategy for Sustainability.
As the premier European programme in the field of exposure to chemicals and health, HBM4EU sees the Strategy as a major opportunity to move forward with the protection of European citizens. We believe that to attain the zero-pollution objective, it is critical to apply tools to monitor the chemical body burden of European citizens and assess the associated health impacts.
To achieve such an ambitious goal, a sustainable surveillance system is needed, a system embedded in legislation that can be used to measure the chemical burden through human biomonitoring. This will support sustainable risk assessment and chemical management in Europe to the benefit of current and future generations.
HBM4EU has established a strong link between science and policy through close interactions with policy makers. Hence, our research and innovation programme has been developed to answer concrete short-term and long-term policy relevant questions from policy makers at European and national levels, and to foster the use of human biomonitoring data in exposure and risk assessment. More precisely, HBM4EU activities are building evidence on exposure pathways, elaborating reference values for exposure and deriving health-based guidance values, as well as feeding into approaches for the risk assessment of chemical mixtures and gaining experience with innovative tools and methods.
Three images illustrate the contribution of HBM4EU to chemicals policy.
The Iceberg. In the Chemicals Strategy for Sustainability the iceberg figure is striking. It shows that we know a lot about approximately 500 out of 100,000 chemical substances on the market, but either little or nothing on most of the others. Obviously, we should continue working on the chemicals of highest concern in terms of risks to health and this is what we do under HBM4EU. However, we are also involved in research on emerging substances using new screening methods, techniques that we are working to harmonize across Europe. The number of substances for which we have little knowledge is so substantial that only a sustained effort over time will allow us to face such a challenge.

The cocktail. One of the major issues addressed in the Strategy is how to identify the most relevant mixtures of chemicals and how to address their impacts. HBM4EU has been carrying relevant research and contributing to working groups on risk assessment and management. Again, this is not an easy matter and long-term research is needed. Yet, there is an urgency to act and, based on the available knowledge, decision makers can already draw some conclusions and act.
The orchestra. The objectives of chemical policy and of HBM4EU cannot be achieved by a single discipline. We need epidemiologists, exposure experts, public health specialists, toxicologists, computational scientists and analytical chemists. This multi-disciplinary collaboration is happening under HBM4EU and in many other EU projects in this field. But the “HBM4EU orchestra” also refers to the number of countries that are part of this programme. Inclusiveness and capacity building are among the main drivers of HBM4EU. While this demands time and investment in the short-term, it is certainly more productive in the long-term, and in line with the European spirit.
If we do not act – with increasing knowledge through research being one way to act – and build our capacities for the long term, we may face some dire consequences in the future. The good news is that HBM4EU partners, together with other national authorities and European Union institutions, are collaborating to develop a future European Partnership for the Assessment of Risk from Chemicals (PARC) to be implemented under the European Union’s next research and innovation framework programme, Horizon Europe.
This partnership will build on the HBM4EU legacy and enlarge its scope. It will bring together European risk assessment and regulatory agencies, as well as policy makers, academia and stakeholders to set a joint research and innovation agenda. This agenda will support EU and national chemical risk assessment and risk management bodies with new data, knowledge, methods, networks and skills to address current, emerging and novel chemical safety challenges. It will facilitate the transition to next generation risk assessment to better protect human health and the environment, in line with the zero-pollution ambition, and will be an enabler for the EU Chemicals Strategy for sustainability.
The Strategy represents the first step towards a zero-pollution ambition for a toxic-free environment. The zero-pollution agenda should start from an understanding of how European citizens are exposed to synthetic chemicals and how these accumulated in the body and make the reduction of the chemical body burden and associated health impacts a key priority. In practice, this can only be delivered through a surveillance system for measuring the exposure burden of environmental pollutants in the European population that is embedded in European Union legislation.
HBM4EU input to policy agenda
HBM4EU has provided direct input to public consultations on the development of strategies and action plans led by the European Commission under the European Green Deal. Follow the links in the table below to access HBM4EU input.
Information Platform for Chemical Monitoring – state of play
Maurice Whelan, Head of Unit on Chemical Safety and Alternative Methods, and Stephanie Bopp, Joint Research Centre, European Commission
The Information Platform for Chemical Monitoring – IPCHEM – provides a wealth of occurrence data on chemicals present in our environment, food, indoor air, and even in our bodies. A recent report published by the European Commission’s Joint Research Centre (JRC) describes how IPCHEM can help us to better understand chemical exposure and support delivery of the key objectives of the Chemicals Strategy for Sustainability. In particular, the strategy envisages a common open data platform to facilitate the sharing and re-use of information on chemicals, including the chemical occurrence data which IPCHEM provides.

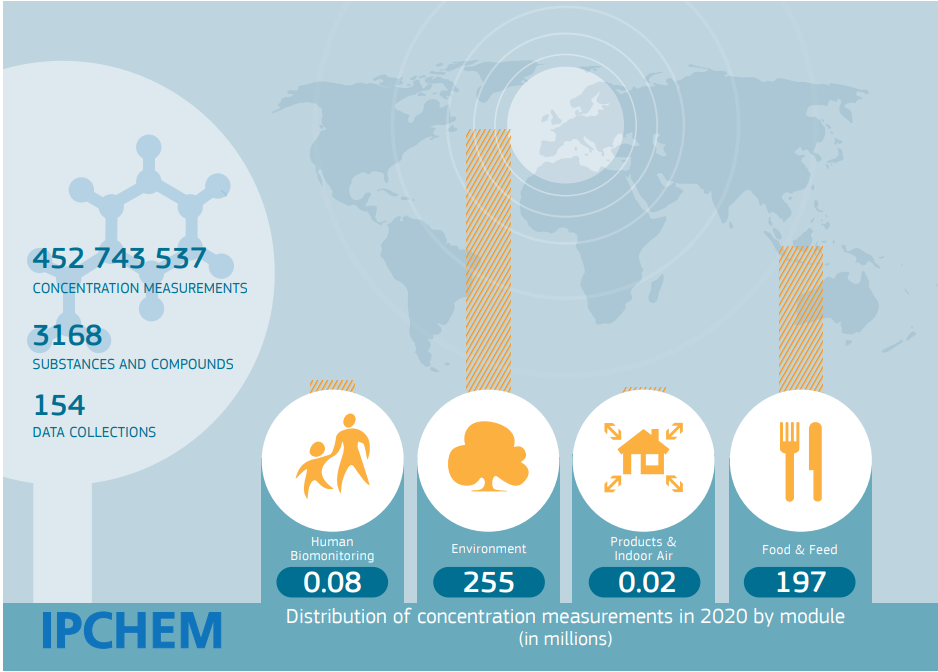
IPCHEM has modules dedicated to the occurrence of chemicals in food, environmental media and indoor air and provides free access to over 150 data collections containing more than 450 million data points. It also includes a module for human biomonitoring data, providing measurements of chemicals in human samples of urine, hair and breastmilk and blood of people of a range of ages, including in the cord blood of new-borns.
The IPCHEM 2020 report provides an overview of recent achievements and describes how its four modules — environment, food and feed, indoor air, and human biomonitoring — are evolving.
Synergies between HBM4EU and IPCHEM
The report includes testimonials from IPCHEM collaborators, including Eva Govarts from VITO, the leader of HBM4EU’s work package on data management and analysis. The human biomonitoring module gained considerable momentum through collaboration with HBM4EU focused on exploiting synergies. As the module coordinator for the human biomonitoring module and an HBM4EU partner, the European Environment Agency has worked to foster these linkages. In particular, VITO played a critical role in facilitating the harmonisation of metadata and data for both HBM4EU and the IPCHEM human biomonitoring module. Metadata has been gathered for more than 120 human biomonitoring data collections throughout Europe and integrated into IPCHEM to make the data collections findable. Aggregated human biomonitoring data for 60 of those data collections were integrated in IPCHEM and are now becoming available to the HBM4EU project partners. In the future datasets will become publicly available, in cases where data providers agree.
Some research questions require the use of individual level data which are protected as personal data under data protection regulations. IPCHEM is preparing hosting facilities and data protection approaches to be able to host also individual level data in the future.
A growing community
IPCHEM is a single access point where multiple stakeholders can find and share information about where chemicals are found and at which concentrations.
Under the scientific and technical leadership of the JRC, IPCHEM is run in collaboration with many different partners across the European Commission, European Agencies, Member State authorities, research consortia, data providers and data users. IPCHEM has gained the trust of regulatory authorities and is now the recommended tool for Member States reporting chemical monitoring data under the Regulation on Persistent Organic Pollutants.
Efforts are ongoing to further populate IPCHEM with new datasets to increase the geographical coverage and the range of chemicals for which data is available in different media. Furthermore, the JRC is developing use cases that illustrate how IPCHEM can help to address questions in the chemical, environmental and health policy areas.
2020 conference on human biomonitoring for science and chemical policy
The international conference on Human Biomonitoring for Science and Chemical Policy took place on October 2nd 2020, in Berlin, under the auspices of the German Presidency of the Council of the European Union (EU).
Representatives from research, administration, industry and civil society met to discuss human biomonitoring and its significance for European chemicals policy.
The conference was opened by Svenja Schulze, the German Minister for the Environment, Nature Conservation and Nuclear Safety. The minister highlighted the many positive initiatives to enhance human biomonitoring, both on the domestic and at EU level, as an important tool for chemical policy making in a context where reliable EU-wide data on the exposure of the population to chemicals are still scarce.

During the conference many speakers, including Virginijus Sinkevičius, EU Commissioner for the Environment, Oceans and Fisheries, and Hans Bruyninckx, Executive Director of the European Environment Agency, emphasised the importance of human biomonitoring for a progressive, sustainable chemicals policy, as an important element of the European Commission’s European Green Deal. This was echoed by Lilian Busse, who spoke on behalf of Dirk Messner, President of the German Environment Agency.
The conference was jointly organised by the Federal Ministry for the Environment, Nature Conservation and Nuclear Safety and the German Environment Agency. Due to Covid-19 restrictions it took place in a hybrid format, with some participants attending in person and others joining online.
All contributions to the conference and some impressions are available on the conference website: www.hbmc2020.com
Recent publications at a glance
 Large-scale suspect and non-targeted screening (NTS) approaches based on high-resolution mass spectrometry (HRMS) are today available for chemical profiling and holistic characterisation of biological samples. These advanced techniques allow the simultaneous detection of a large number of chemical features, including markers of human chemical exposure. Such markers are of interest for biomonitoring, environmental health studies and support to risk assessment. Furthermore, these screening approaches have the promising capability to detect chemicals of emerging concern (CECs), and provide early warning support to policy. Whilst of growing maturity in the environment and food safety areas, NTS remains poorly addressed in the field of human biomonitoring. The main aim of this paper was to present an overview of the basic principles, promises and challenges of suspect and non-targeted screening approaches applied to human samples.
Large-scale suspect and non-targeted screening (NTS) approaches based on high-resolution mass spectrometry (HRMS) are today available for chemical profiling and holistic characterisation of biological samples. These advanced techniques allow the simultaneous detection of a large number of chemical features, including markers of human chemical exposure. Such markers are of interest for biomonitoring, environmental health studies and support to risk assessment. Furthermore, these screening approaches have the promising capability to detect chemicals of emerging concern (CECs), and provide early warning support to policy. Whilst of growing maturity in the environment and food safety areas, NTS remains poorly addressed in the field of human biomonitoring. The main aim of this paper was to present an overview of the basic principles, promises and challenges of suspect and non-targeted screening approaches applied to human samples.
Jean-Philippe AntignacPeer-reviewed articles: Pourchet M, Debrauwer L, Klanova J, Price EJ, Covaci A, Caballero-Casero N, Oberacher H, Lamoree M, Damont AL, Fenaille F, Vlaanderen J, Meijer J, Krauss M, Sarigiannis D, Barouki B, Le Bizec B, Antignac JP. Suspect and non-targeted screening of chemicals of emerging concern for human biomonitoring, environmental health studies and support to risk assessment: from promises to challenges and harmonisation issues. Environment International 2020;139:105545. https://doi.org/10.1016/j.envint.2020.105545.
This publication developed under HBM4EU is evaluating needs and lessons learned from projects in the domains of public and environmental health and their benefit of exchange for better health promotion. HBM4EU and BRIDGE Health are important EU-projects in these fields and their experiences showed that continuous efforts must be undertaken, also by succeeding projects, to guarantee the exchange between public health and environmental health issues. Networks covering both are essential to provide better evidence of knowledge. Within this publication the need for focused exchanges and collaborations between the projects were identified and benefits of exchanges were highlighted for the specific areas of indicator development, linkage of data repositories and the combination of HBM studies and health examination surveys (HES). Further recommendations for a European wide harmonisation among different tasks in the fields of public health and environmental health are being developed.
The experiences from the projects BRIDGE Health and HBM4EU give a valuable input for any future activity in these domains. This publication shows, that avoiding overlaps and streamlining further exchange of public health and environmental health contributes to best use of research results and allows to develop new strategies and tools for improvement of health information and thus enhances people’s health and well-being.
Madlen David, German Environment Agency, Berlin
David, M., Schwedler, G., Reiber, L. et al. Learning from previous work and finding synergies in the domains of public and environmental health: EU-funded projects BRIDGE Health and HBM4EU. Arch Public Health 78, 78 (2020). https://doi.org/10.1186/s13690-020-00460-9
The paper was accepted with minor changes, for the special issue in the International Journal of Hygiene and Environmental Health and it is in the revision process. The below is a brief note informing about the end of the QA/QC programme for all the target substances in HBM4EU and the current status of the analytical phase.
The HBM4EU project faces the final stretch and after huge efforts and some unexpected problems due to covid-19, great results are coming. An example is the completion of the QA/QC programme of WP9 that can be considered as a great success within the HBM4EU challenges. In parallel, the analytical phase has been moving forward and in November 2020, the 58 % of the analysis were completed and 24% ongoing (18% of the samples not received in the laboratories yet). The main results will be presented in the publication, including some graphs for a more impressive overview of the achievements.
Stefanie Nübler, Marta Esteban López, Argelia Castaño, Hans Mol, Moritz Schäfer, Karin Haji-Abbas-Zarrabi, Daniel Bury, Holger M. Koch, Vincent Vaccher, Jean-Philippe Antignac, Darina Dvorakova, Jana Hajslova, Cathrine Thomsen, Katrin Vorkamp, Thomas Göen. Interlaboratory Comparison Investigations (ICI) and External Quality Assurance Schemes (EQUAS) for cadmium in urine and blood: results from the HBM4EU project (accepted with minor changes, for the special issue of International Journal of Hygiene and Environmental Health).
Disclaimer
The HBM4EU project was launched in 2016 with the aim of improving the collective understanding of human exposure to hazardous chemicals and developing HBM as an exposure assessment method. The project had €74m in funding and jointly implemented by 120 partners from 28 participating countries – 24 EU member states plus Norway, Switzerland, Iceland and Israel and the European Environment Agency. One of its aims was to ensure the sustainability of HBM in the EU beyond 2021. The project ended in June 2022. The website will not be updated any longer, except the page on peer reviewed publications, but will be online until 2032.